Introduction
Last week I was working with a Chemist, doing a crystalline study on a group of metal ores. We went back and forth between the pXRD and the single crystal XRD to determine the crystalline structure of the different metal ores throughout the veins. We measured the lattice, analyzed the bonds, and weighed the electrons. The chemist continually commented on how the lattice looked like a nitrogen bond. Nitrogen is hard to test for in rocks. The largest nitrogen reserves in the rock record are found in hydrocarbon deposits. The nitrogen content in oil is correlated to the sulfur content. For the sake of simplicity, I’ll say that sweat crudes have ~0.1-0.5% nitrogen and sour crudes have ~0.5-1%. Sweat crudes have a sulfur content of <0.5%, and sour crudes >0.5%.
During refining in the USA, virtually all the nitrogen and sulfur are removed. US refiners do an ok job reducing the nitrogen and sulfur in their products. For example, suppose the oil comes from the ground with 0.5% sulfur equaling 5000 ppm (parts per million). The low-sulfur diesel in your truck has 15 ppm and the gasoline 10 ppm.
Nitrogen Cycle
Nitrogen Fixation
Nitrogen fixation is the process by which nitrogen is removed from the atmosphere and converted into ammonia (mainly) to support plant and animal life. Bacteria in soil, ocean, and plants convert atmospheric nitrogen (78% of the air we breathe) to ammonia for use by plants and animals to sustain life.
Plants and animals use ammonia (NH3) to make amino acids, proteins, nucleic acids, and chlorophyll.
Chlorophyll
Chlorophyll enables the transformation of light energy into chemical energy in plants.
There are six types of Chlorophyll, A, B, C, D, E & F.
Each type of chlorophyll contains a Chlorin ring, a ring of four nitrogen atoms bound to an atom of magnesium in the center.
Protein and Amino Acids
All amino acids comprise carbon, hydrogen, nitrogen, and oxygen.
All protein is built from the twenty amino acids listed below with peptide bonds. For a peptide bond to form between the two amino acids, one hydrogen atom and two oxygen atoms need to be removed.
Nucleic Acids
The three main types of nucleic acids are Deoxyribonucleic acid (DNA), Ribonucleic acid (RNA), Artificial nucleic acid, and Peptide nucleic acid (PNA). DNA and RNA are found in the cell nuclei. Artificial nucleic acid and PNA are methods used in bioscience to modify DNA/ RNA. Artificial nucleic alters the sugar and/ or the phosphate backbone. PNA uses a nitrogen-based molecule like an amino acid to modify DNA/ RNA instead of sugar/phosphoric acid. DNA/ RNA are composed of a sugar or derivative of a sugar, a nucleobase (cytosine, guanine, adenine, thymine, or uracil), and phosphoric acid. The DNA nucleobase contains cytosine, guanine, adenine, and thymine, while RNA contains uracil, cytosine, guanine, and adenine. Each part of the nucleobase in DNA and RNA is a nitrogen-based chemical.
Nitrogen in Rocks
As previously mentioned, soil and ocean bacteria take nitrogen from the atmosphere and turn it into ammonia. Over the geologic timescale, some ammonia makes it into the rock cycle. An example of this would be nitrogen salt deposits. Caliche in Chile contains large amounts of (sodium nitrite) saltpeter used to manufacture gunpowder. During the US Civil War, Confederate and Union forces fought over caves of bat guano from Texas to Virginia because they needed the sodium nitrite in the feces to manufacture gunpowder. During the First World War, the search for nitrogen expanded, and field tests were developed to hunt for nitrogen-rich sediments across the USA. Only a few were found. Over the last few years, climate modelers have become more aware of nitrogen gas trapped in rocks, which has implications for climate models. Gold exploration is now using nitrogen petrogenesis for hunting for gold resources. Small pockets of nitrogen gas trapped in rocks remain inert during metamorphism. Gold explorers can then trace the nitrogen backward and search for the source rock.
Why Does the Earth’s Nitrogen System Matter So Much?
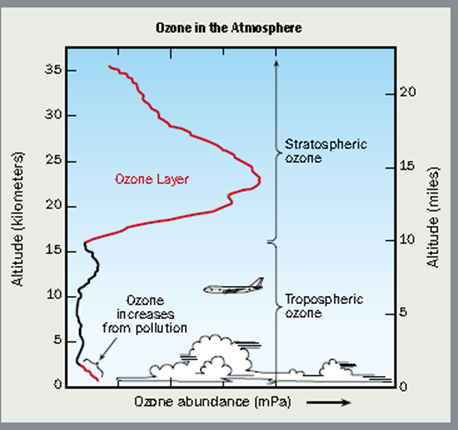
A strong and vibrant nitrogen cycle is needed for life to increase and thrive on Earth. We must think about how to help the Earth succeed and support 40-50 billion people. Burning fossil fuels is the primary source of new nitrogen in the atmosphere. While we support increased CO2 emissions up to 1500 ppm, currently around 400 ppm globally, we need to rethink nitric oxide emissions. When nitrogen is burnt inside a combustion engine, it mixes with oxygen and forms nitric oxides. Nitric oxides are sensitive to ultraviolet light and are split apart by the sun. When broken apart, nitrogen is released, which improves plant life, but the oxygen portion bonds together to form O3, which is ozone. Ozone is one of the most essential parts of our atmosphere. Ozone in the Earth’s stratosphere protects the planet from 98 percent of the sun’s ultraviolet radiation and sits 20-30 km above the Earth’s surface. The ozone from fossil fuels stays below 5km and is the primary constituent of smog. Smog causes allergies and other health problems, but it does the same job below 5km as it does between 20-30km; it blocks UV.
Four Part Solution
One: Aggressively improve relations with South Africa. South Africa has the world's most significant production and highest reserves of platinum metal group elements (iridium, osmium, palladium, platinum, rhodium, and ruthenium). Palladium, platinum, and rhodium are used to make catalytic converters. Catalytic converters break the nitric oxide bond without UV light. What is supposed to come out of your tailpipe is nitrogen gas and oxygen (O2).
Two: Governments should subsidize catalytic converters. Catalytic converters are expensive. The government should mandate and subsidize new catalytic converters for all emission sources every five years. As combustion vehicles age, catalytic converters lose capacity.
Three: Instead of using catalytic converters on industrial sources, create new technology that mimics the UV splitting process. Release the nitrogen into the lower atmosphere and inject the ozone into the ozone layer 20-30km above the earth’s surface where it should be. Don’t just stop it from shrinking; we must actively grow the ozone layer.
Four: Continue supporting catalytic technology research and speed up the adoption. Example:
https://chemistry.harvard.edu/news/cooler-catalytic-converters-cleaner-air-all